We start with our balanced chemical reaction, which because we are looking at a precipitation reaction is: Now we simply need to sum up the AG's for the product symbol means the sum up): minus the sum of AG's for the reactants (the Σ EAG f(ractants) AGn +184.01 kJl/mol)-(-79.31 kl/mol+ 111.34 kJ/mol) +6.64 kJ/mol As expected! Since we knew that the dissolving of ammonium nitrate was spontaneous at room temperature and and standard state concentration, we also knew it would not precipitate out of solution at room temperature da ith the reagents which are not at standard state condition, the value of ΔG standard state concentration. Ter's cheek the change in Gibbs free energy for the precipitation of ammonium nitrate u of formation from the table above. Therefore, we must write the balanced chemical equation for a precipitation reaction (that 1S, the opposite of dissolving)-for precipitation reactions, the solid is always written as a product (the opposite of a solubility problem). 
However, we are interested in the determining whether or not a pair of reagents will precipitate. The table above listed the standard state changes in Gibbs free energy for the components in the reaction of ammonium nitrate dissolving. You will use the calculated values to decide whether a precipitate should form when two solutions are mixed. 
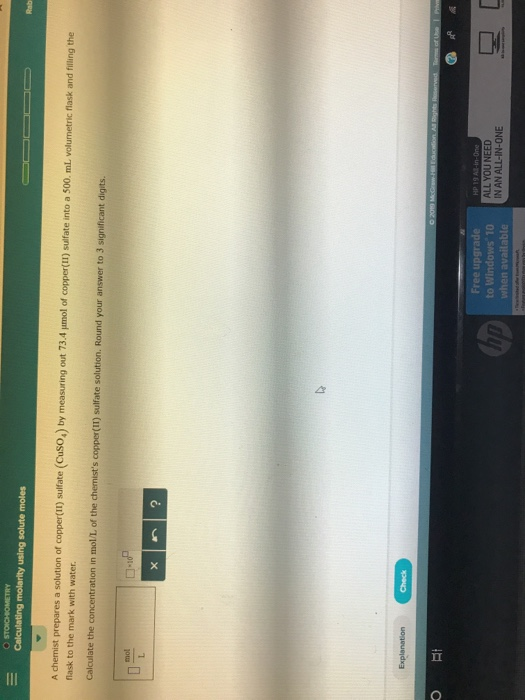
How to Calculate the Free Energy of a Reaction, AGr In today's experiment, you are going to calculate the AGrn for a variety of possible precipitation reactions (the subscript rxn -for reaction-is no different that the f-for formation just that rxn is more general). That is, ammoniunm nitrate will spontaneously dissolve in water at room temperature even though the reaction is endothermic. The negative sign on the final answer tells us that this would be a spontancous reaction. 6.01 kJ/mol NOTE THE UNITS HERE because Gibb's free energy is typically given in kJ/mol, I went ahead and converted the units on the entropy into kJ/mol K. NOTE: The units on entropy are typically given in Joules per mole Kelvin, while the units on enthalpy are given in units of kilojoule per mole! You must pay attention to what you are doing to keep the units straight! For enthalpy we have: ΔΗ, (-132.51 kJ/mol +-206.65 kJ/mol)-(-365.56 kJ/mol)-+26.40 kJ/mol The change in entropy then is AS-(113.40 J/mol K +146.40 J/mol K)-(151.08 J/mol K) +108.72 J/mol K Since the standard state is 298.15 K, the change in free energy is: Δ0,". Solution: We start where we always start, with the balanced chemical equation: is Species Name Enthalpy "AH" (k.J/mol) -365.56 -132.51 -206.65 Entropy "S" (J/mol K) 151.08 113.40 146.40 Free energy "AGf (k.J/mol) -184.01 -79.31 -111.34 NH NO (s) NH4 NO Using the table above, we can calculate the change in enthalpy and entropy using the standard products minus reactants (multiplied by the coefficient from the balanced chemical equation). Let's calculate ΔΗ% AS, and ΔG f for the dissolving of ammonium nitrate to determine whether the reaction spontancous or not at standard state conditions. Transcribed image text: 175 Data Sheet: Thermo: Predicting Precipitation Reactions Name: Partner Solutions Possible Precipitate onmala or #2 W2 #1 #1 #2 oNalAgNO, Nal BaCl トse- N2 BCAgNO Agel BaCk Na,SO, BaC, Ca(NO,h ト)R NaSOAgNO, NaNO SO4 Ca(NO,h 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
